|
Bohr's model and postulates do not explain this phenomenon. Spectral lines undergo splitting when a sample of gas is affected by an electric field (Stark effect) and magnetic field (Zeeman effect).These are referred to as hyperfine spectral lines. Bohr's model does not explain the presence of hyperfine lines. Each spectral line, when examined at higher magnification, actually consists of a number of smaller fine lines.The accuracy decreases as the effective nuclear charge of an atom or ion increases (due to greater number of protons). Predictions made by Bohr’s model is only accurate for the hydrogen atom. 
Bohr's model cannot predict the relative intensity (brightness) of the spectral lines.In other words, his model does not explain why electrons can remain in their orbits without spiralling into the nucleus due to electrostatic attraction. Bohr does not provide an explanation to 'stationary states' of electrons.Circular motion of electrons is from classical physics while the quantisation of its momentum and energy of orbits is from quantum physics. Bohr's model combines principles from both classical and quantum physics.$$\Delta E_ J$$ Limitations of Bohr's Atomic Modelīohr's model of the atom has several limitations. Niels Bohr, Danish physicist, used the planetary model of the atom to explain the atomic spectrum and size of the hydrogen atom. An electron can transition between orbits by absorbing or releasing energy that is exactly equal to the difference in energy of orbits, consistent with the law of conservation of energy.Įlectron excitation occurs when an electron absorbs energy to move to an orbit of higher energy.Įlectron relaxation occurs when an electron moves to a lower orbit, releasing energy in the form of electromagnetic radiation (photon). Bohr’s theory explained the atomic spectrum of hydrogen and established new and broadly applicable principles in quantum mechanics. 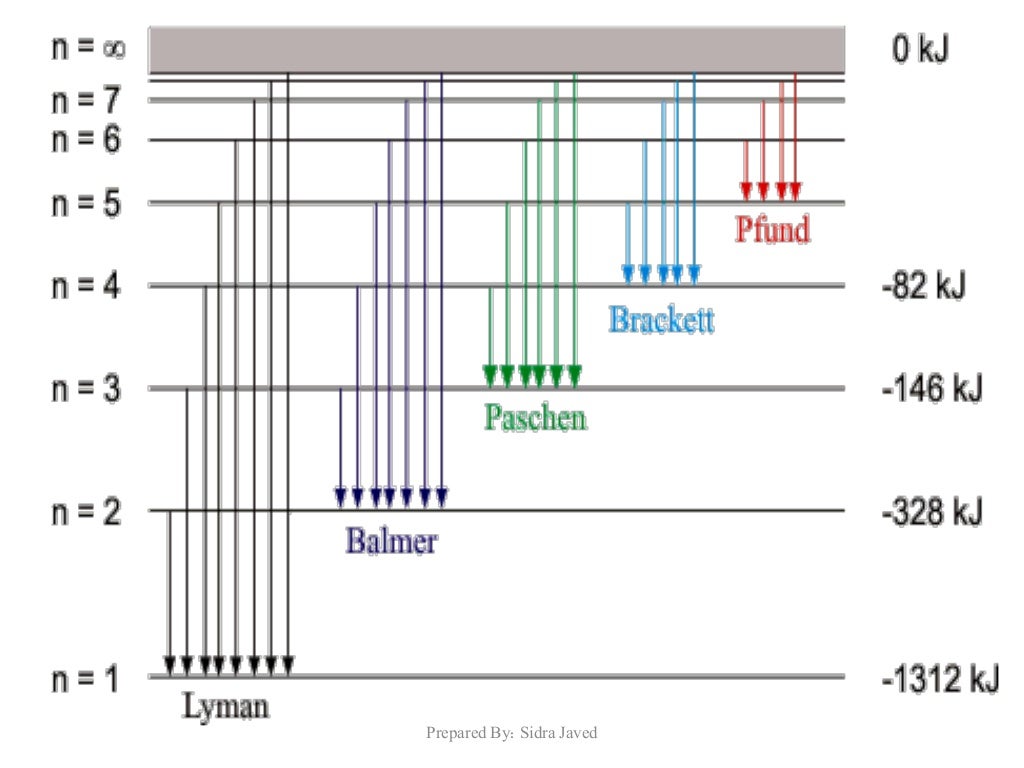
In these orbits, electrons exist in 'stationary states' and do not emit energy.īohr’s model of the atom describes electrons orbiting in stable energy levels as opposed to Rutherford's model in which electrons' motion was not described.Ģ. Electrons revolve around the nucleus in circular orbits with discrete radii and quantised energies. Niels Bohr proposed three postulates in his atomic model:ġ. – Rydberg's equation Bohr's Model of the Atom The Balmer series, or Balmer lines in atomic physics, is one of a set of six named series describing the spectral line emissions of the hydrogen atom. With only one electron and one proton hydrogen is the simplest atom and early studies of its line spectra lead to some of the first theories with an experimental foundation for our understanding of the atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
